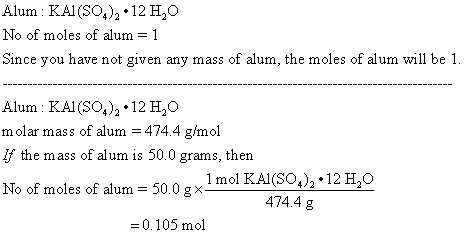Kal(so4)2.12h2o Aluminium Potassium Sulfate Potassium Alum Powder - Buy Potassium Alum Powder,Aluminium Potassium Sulfate Powder,Kal(so4)2 12h2o Potassium Alum Product on Alibaba.com

Kal(so4)2 12h2o Aluminum Potassium Sulfate Lump Potassium Alum Potash Alum - Buy Potash Alum,Aluminum Potassium Sulfate,Kal(so4)2 12h2o Potash Alum Product on Alibaba.com
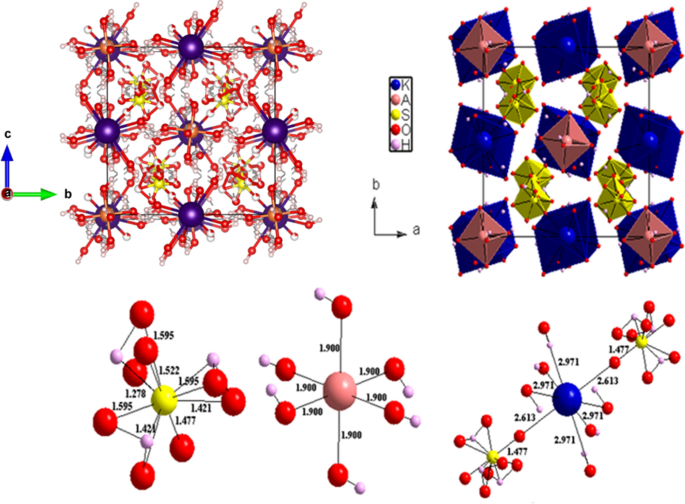
Investigations on KAl(SO4)2∙12H2O: A Candidate α-Alum Material for Energy Storage Applications | SpringerLink

Kal(so4)2.12h2o Aluminium Potassium Sulfate Potassium Alum Powder - Buy Potassium Alum Powder,Aluminium Potassium Sulfate Powder,Kal(so4)2 12h2o Potassium Alum Product on Alibaba.com

Anomalous longitudinal acoustic phonon and elastic constant in potassium alum KAl(SO4)2·12H2O single crystal studied by Brillouin spectroscopy - ScienceDirect
Thermal behaviour of alum-(K) KAl(SO4)2·12H2O from in situ laboratory high-temperature powder X-ray diffraction data: thermal e
![Table I from Alum [KAl(SO4)2·12H2O]: An Efficient, Novel, Clean, Catalyst for Doebner—Knoevenagel Reaction for the Efficient Production of α,β‐Unsaturated Acids. | Semantic Scholar Table I from Alum [KAl(SO4)2·12H2O]: An Efficient, Novel, Clean, Catalyst for Doebner—Knoevenagel Reaction for the Efficient Production of α,β‐Unsaturated Acids. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f3ee51bfb8cf7b09279205adbb73911287275c36/2-TableI-1.png)
Table I from Alum [KAl(SO4)2·12H2O]: An Efficient, Novel, Clean, Catalyst for Doebner—Knoevenagel Reaction for the Efficient Production of α,β‐Unsaturated Acids. | Semantic Scholar

Suppose Harry begins with the hydrate KAl(SO4)2·12H2O. After dehydration he finds that he is left with 3.0 - Brainly.com